Lesson Notes: GASES
1. Laboratory Preparation of Carbon Dioxide and Ammonia and Their Properties
1.1 Preparation of Carbon Dioxide (CO₂) in the Laboratory
Principle:
A metal carbonate reacts with a dilute acid to produce CO₂ gas.
Common Method:
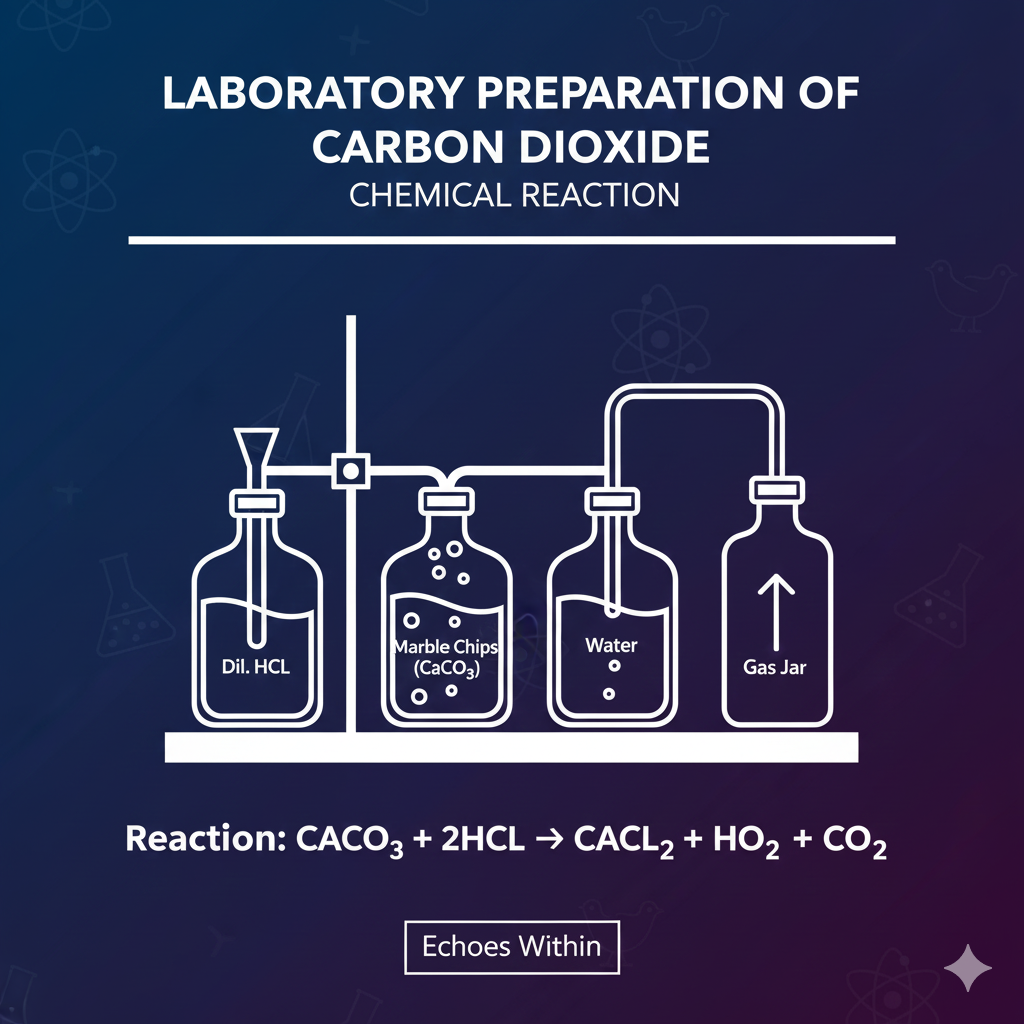
-
Chemical Reaction:
-
Apparatus: Conical flask, thistle funnel, delivery tube, gas jar.
-
Procedure:
-
Put marble chips (CaCO₃) in the conical flask.
-
Add dilute HCl through the thistle funnel.
-
CO₂ is produced and collected by downward delivery (since CO₂ is heavier than air).
-
1.2 Properties of Carbon Dioxide
-
Physical Properties:
-
Colourless and odourless gas
-
Slightly soluble in water
-
Denser than air
-
Turns lime water milky (forms insoluble CaCO₃)
-
-
Chemical Properties:
-
Non-combustible (does not burn)
-
Does not support burning
-
Reacts with metal oxides to form carbonates
-
Forms carbonic acid when dissolved in water
-
Practice : MCQs – Carbon Dioxide (CO₂)
1.3 Preparation of Ammonia (NH₃) in the Laboratory
Principle:
A mixture of ammonium salt and a strong alkali is heated to produce ammonia gas.
Common Reaction:
Procedure:
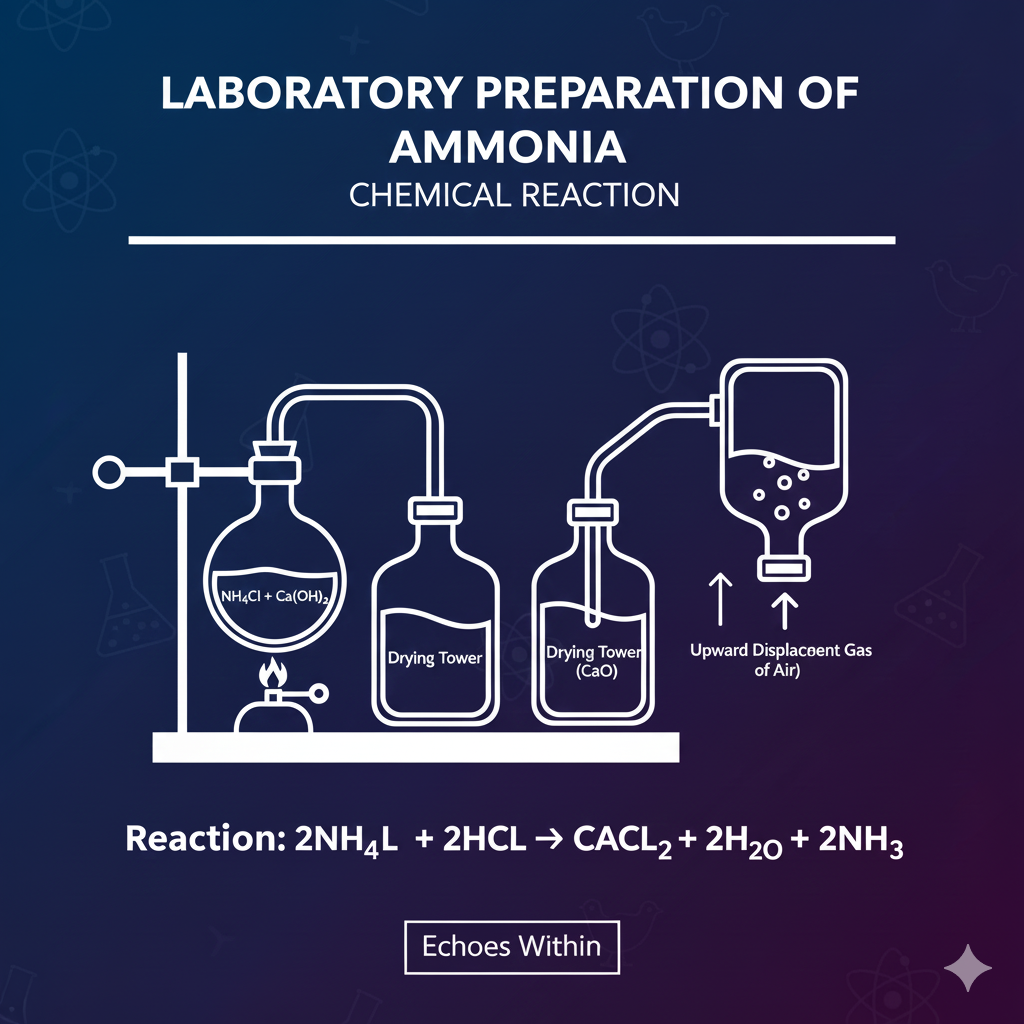
-
A mixture of ammonium chloride and slaked lime is heated in a flask.
-
Ammonia gas is formed and collected by upward delivery (lighter than air).
-
Use drying agent CaO to remove moisture (NH₃ dissolves in water).
1.4 Properties of Ammonia
-
Physical Properties:
-
Colourless gas with a sharp, pungent smell
-
Extremely soluble in water (forms ammonium hydroxide)
-
Lighter than air
-
-
Chemical Properties:
-
Basic in nature
-
Turns red litmus blue
-
Combusts in oxygen to give nitrogen and water
-
Reacts with HCl gas to produce dense white fumes of ammonium chloride
-
2. Importance / Utility of Carbon Dioxide and Ammonia
2.1 Uses of Carbon Dioxide
-
Used in fire extinguishers
-
Used in carbonated beverages (cold drinks)
-
Dry ice (solid CO₂) is used for refrigeration
-
Essential for photosynthesis in plants
-
Used in welding and chemical industries
2.2 Uses of Ammonia
-
Used in manufacturing fertilisers (urea, ammonium sulfate)
-
Used in the production of nitric acid
-
Used as a refrigerant
-
Cleaning agent in households (ammonium hydroxide)
-
Used in the textile, plastic, and pharmaceutical industries
Practice :MCQs – Ammonia (NH₃)
3. Acid Rain: Causes, Effects, and Control Measures
3.1 Causes of Acid Rain
Acid rain forms when acidic gases dissolve in rainwater.
Major pollutants:
-
Sulfur dioxide (SO₂) from industries, brick kilns, and fossil fuel burning
-
Nitrogen oxides (NOₓ) from vehicles, power plants
-
Carbon dioxide (CO₂) contributes weakly to acidity
These gases react with water vapour to form:
-
Sulfuric acid (H₂SO₄)
-
Nitric acid (HNO₃)
3.2 Effects of Acid Rain
-
Damages buildings (especially marble monuments)
-
Reduces soil fertility
-
Harms aquatic ecosystems (fish die due to pH change)
-
Affects plant growth
-
Corrodes metals and infrastructure
-
Causes respiratory problems in humans
3.3 Control Measures
-
Use low-sulfur fuels
-
Install scrubbers in industries and power plants
-
Promote electric vehicles and public transport
-
Use renewable energy sources
-
Reforestation and afforestation
-
Strict emission regulations
4. Greenhouse Effect: Causes, Consequences, and Control Measures
4.1 Causes of the Greenhouse Effect
The greenhouse effect occurs due to excessive greenhouse gases in the atmosphere.
Major greenhouse gases:
-
Carbon dioxide (CO₂)
-
Methane (CH₄)
-
Nitrous oxide (N₂O)
-
Water vapor
-
CFCs (Chlorofluorocarbons)
Human activities that increase greenhouse gases:
-
Burning fossil fuels
-
Deforestation
-
Industrial pollution
-
Excessive agriculture (cattle farming emits methane)
-
Use of refrigerators and ACs (CFC leakage)
4.2 Consequences / Effects of the Greenhouse Effect
-
Global warming
-
Melting of glaciers and rising sea levels
-
Unpredictable weather and climate change
-
Increase in natural disasters (floods, droughts, cyclones)
-
Extinction of species
-
Reduced agricultural productivity
4.3 Measures to Minimise Greenhouse Effect
-
Reduce fossil fuel use
-
Promote solar, wind, and hydropower
-
Plant more trees (afforestation)
-
Use energy-efficient appliances
-
Reduce, reuse, recycle
-
Minimise the use of CFC-based products
-
Practice sustainable agriculture
Practice :20 MCQs – Whole Lesson on Gases
Conclusion (SEO-Friendly)
Understanding the preparation, properties, and uses of gases like carbon dioxide and ammonia helps students connect chemistry with everyday life. Likewise, learning about acid rain and the greenhouse effect builds environmental awareness and encourages sustainable practices. These notes provide clear, exam-focused explanations for NEB/SEE students.
Practice: ⭐ HOTS MCQ Set – Gases
