SEE Practice Model Set 2
[EWA1B2C3D0]
Time: 3 hours FM: 75
All the questions are compulsory.
Group ‘A’ [10X1=10]
1. Write the best alternative to the following questions in the answer sheet.
a. Which is the smallest unit of computer memory?
I. Kilobyte
ii. Megabyte
iii. Bit
iv. Gigabyte
b. Why is pine kept in the sub-division gymnosperm?
I. It has needle-shaped leaves.
ii. It has naked seed.
c. Which of the following groups of organism are closely related on the basis of evolution?
A. Porifera, Annelida, Chordata
B. Porifera, Arthropoda, Chordata
C. Coelenterata, Arthropoda, Chordata
D. Platyhelminthes, Nemathelminthes, Annelida
d. What is the main cause of increasing the level of ocean?
A. Deforestation
B. Heavy rainfall
C. Flood and landslide
D. Increase in the temperature of the earth.
e. Calculate the mass that a person can lift on the Moon if he can lift a mass of 50 kg on the earth. The value of g on the surface of the Earth is 9.8 m/s² and that of the moon is 6 times less.
A. 200.6 kg
B. 300.6 kg
C. 400.6 kg
D. 500.6 kg
f. If large Piston A and small Piston B of a hydraulic lift have their cross-sectional area, pressure and force / weight are A1 and A2, P1 and P2 and F1 and F2 respectively, then which of the following reaction would be correct according to Pascal’s law?
A. P1 > P2
B. A1 <A2
C. F1 > F2
D. F1 < F2
g. Identify the characteristics of the image of the object XY kept in front of the lens as shown in the given figure.
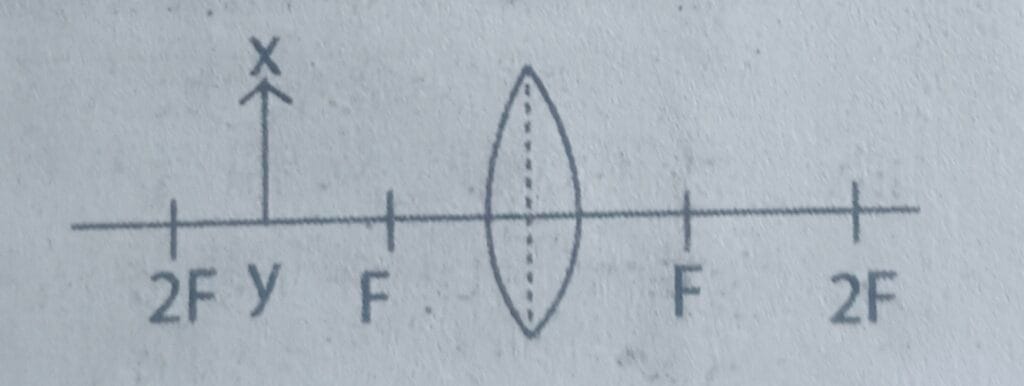
A. Virtual, erect and magnified
B. Real, erect and magnified
C. Virtual, inverted and diminished
D. Real, inverted and magnified
h. Which of the following is proved by Hubble’s law?
A. Universe is contracting
B. University is expanding
C. University remains same
D. Galaxies appear to be closer
I. Which one is a double displacement reaction from the following?
A.
j. A statement and two arguments are given below.
Statement: Detergent is called soapless shop
Argument 1: It has cleaning properties like soap but it’s chemical nature is different.
Argument 2: It does not give lather with hard water as soap.
A. Statement and argument 2 are correct but argument 1 is incorrect
B. Statement and argument 2 are incorrect but argument 1 is correct.
C. Statement and argument 1 are correct but argument 2 is incorrect.
D. Statement and argument 1 are incorrect but argument 2 is correct.
Group ‘B’ [9X1 =9]
Very short answers types questions.
a. What are dependent variables? Write with an example.
b. Write the relation between upthrust and weight of displaced liquid.
c. The apparent depth of water in a pond appears less than the real death. why?
d. What is the focal length of lens having power 1 diopter?
e. A fan in a room rotates when its switch is made on, which principle is used here?
f. Which force is responsible for the existence of heavenly bodies in the universe?
g. Write the name off name aur off iron.
h. Why is reduction of active metal possible only by the electro refining method?
I. Write the molecular formula of the given compound.
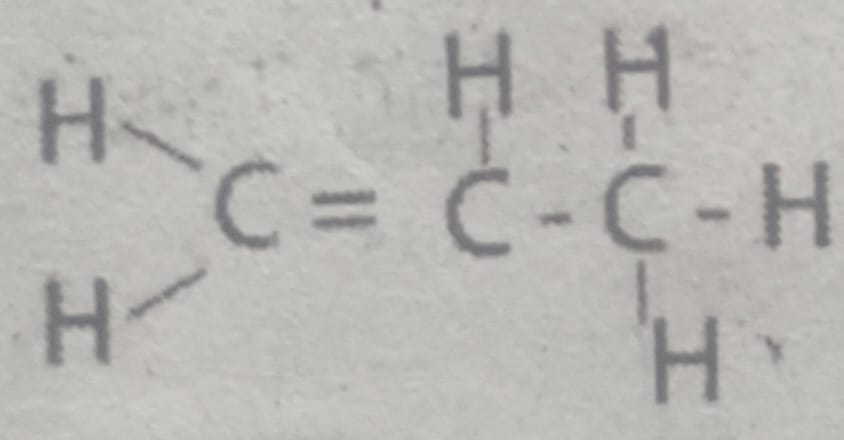
Group ‘C’ [14X2=28]
Short answer type questions.
3. Check the correctness of the equation V² = U²+ 2as² by using unit wise analysis.
4. Write any two characteristics of monocotyledon plant.
5. Study the concept map given below and mention the number of chromosomes in X and Y to distinguish haploid and diploid.
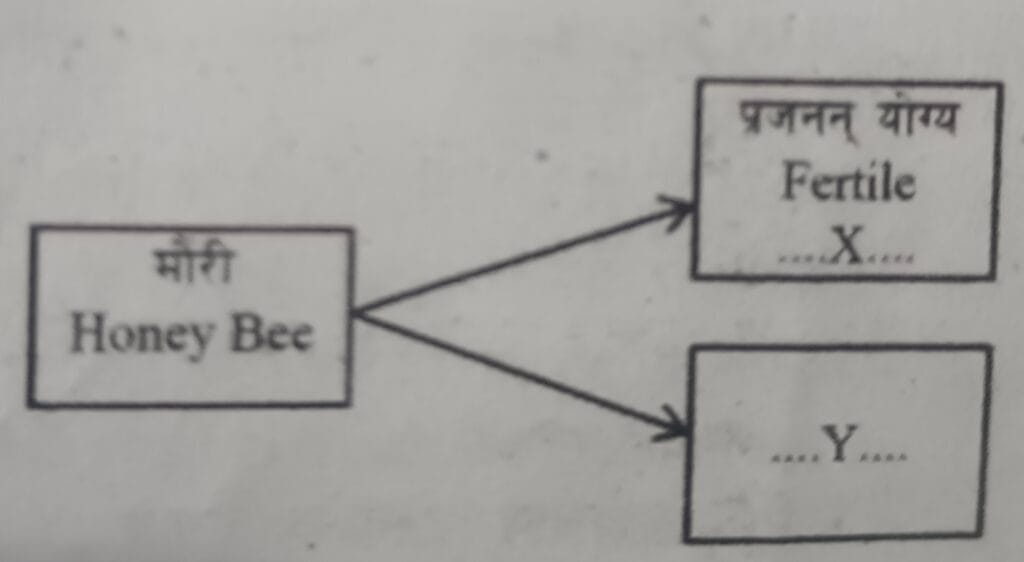
6. Write any two differences between mitosis and meiosis cell division.
7. Draw a probability table of the result obtained in sex determination.
8. Mention two functions of blood on human body.
9. Identify the medicinal plant shown in the figure and write its uses.

10. What is acceleration due to gravity? What is the value of acceleration due to gravity on the pole of the earth?
11. If gravitational force between A and B is F. What is the gravitational force between B and C in terms of F.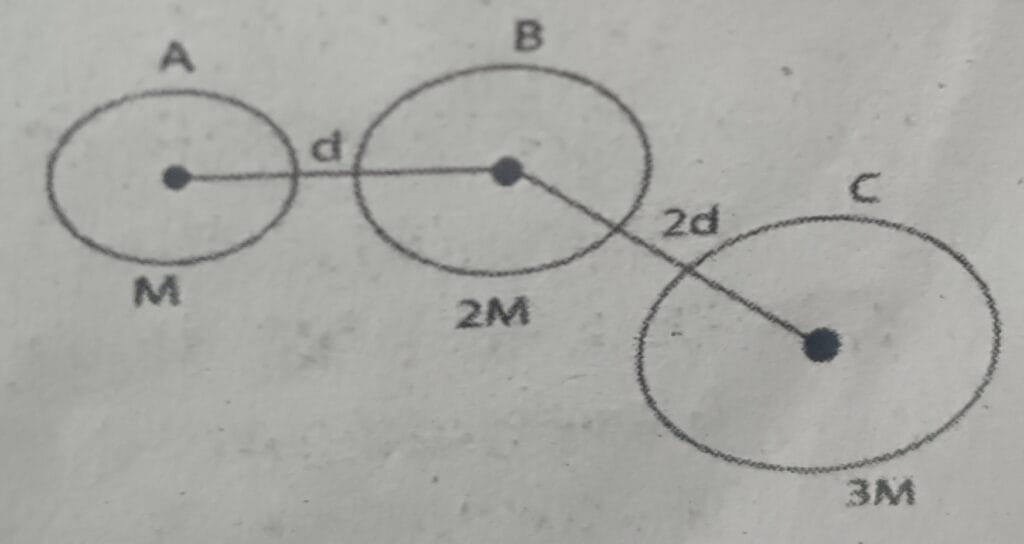
12. Write any two differences between electric motor and generator.
13. If the load transmission at Dhalkewar Dhanusha transmits electricity of 400 KV, then which type of transformer is used here? Also draw a figure showing primary (input) coil and secondary (output) coil in it.
14. A thin zinc plate was kept in a glass container having copper sulphate solution. On examining, it was found that blue colour of solution is getting lighter and lighter. After few days when zinc plate was taken out of the solution a number of small holes where noticed in it. State the reason and write the chemical equation of the reaction.
15. Monohydric alcohol is used as thermometric liquid and as disinfectant in syringe and wounds. On the basis of statement, write the answer of the following questions.
a. Write structural formula of that alcohol.
b. Right molecular formula of 4th member of alcohol of the homologous series in which the given alcohol lies.
16. How is bacterial growth inhibited by salting or adding sugar in food materials?
Group ‘D’
Write long answer. [7 X4 =28]
17. “A blog addressed at https://www.celebree.com/blog/resources/the-pros-and-cons-of-technology-in-education/ writes: …… Technology has become a foundational component in many areas of our daily lives, as well as our childrens. Within the confines of the classroom and learning environment, technology can be both beneficial and problem matric for students. …..”
I. Which technology does the blog describe?
ii. The technology mentioned in the blog also has advantage. Write any two disadvantages.
iii. Mention anyone use of AI in the technology mentioned above.
18. Read the newspaper article and answer the questions that follow.
A copying machine for living bWithout
Without any doubt, if there had been elections for the animal of the Year 1997, Dolly would have been the winner! Dolly is a Scottish ship that you see in the photo. But Dolly is not just a simple sheep. She is a clone of another sheep. A clone means: a copy. Cloning mans copying ‘from a single master copy’.
She is a clone of another sheep. A clone means: a copy. Cloning mans copying ‘from a single master copy’.
It was the Scottish scientist Ian wilmut who designed the ‘copying machine’ for sheep. He took a very small piece from the udder of an adult sheep (sheep 1).
From that small piece he removed the nucleus, then he transferred the nucleus into the egg-cell of another (female) sheep (sheep 2). But first he removed from that egg cell all the material that would have determined sheep 2 characteristics in a lamb produced from that egg-cell.Ian Wilmut implanted the manipulated egg-cell of sheep 2 into yet another (female) sheep (sheep 3). Sheep 3 become pregnant and had a lamb: Dolly.
Some scientists think that within a few years it will be possible to clone people as well. But many governments have already decided to forbid cloning of people by.
I. Which sheep is Dolli identical to?
ii. In the text, part of the udder that was used is described as ‘a very small piece’. From the article text you can work out what is meant by “a very small piece”.
iii. In the last sentence of the article it is stated that many governments have already decided to forbid cloning of people by law. Two possible reasons for this decision are mentioned below. Are these reasons scientific reasons? Circle either ‘Yes’ or ‘No’ for each.
Reasons | Scientific |
Cloned people could be more sensitive to certain diseases then normal people. | Yes / No |
People should not take over the role of a creator. | Yes/No |
19. Write any two symptoms of high blood pressure and any two preventive measures. (K)
20. The relation between volume of water and temperature is shown in the given graph. Study the graph and answer the following question. (HA)
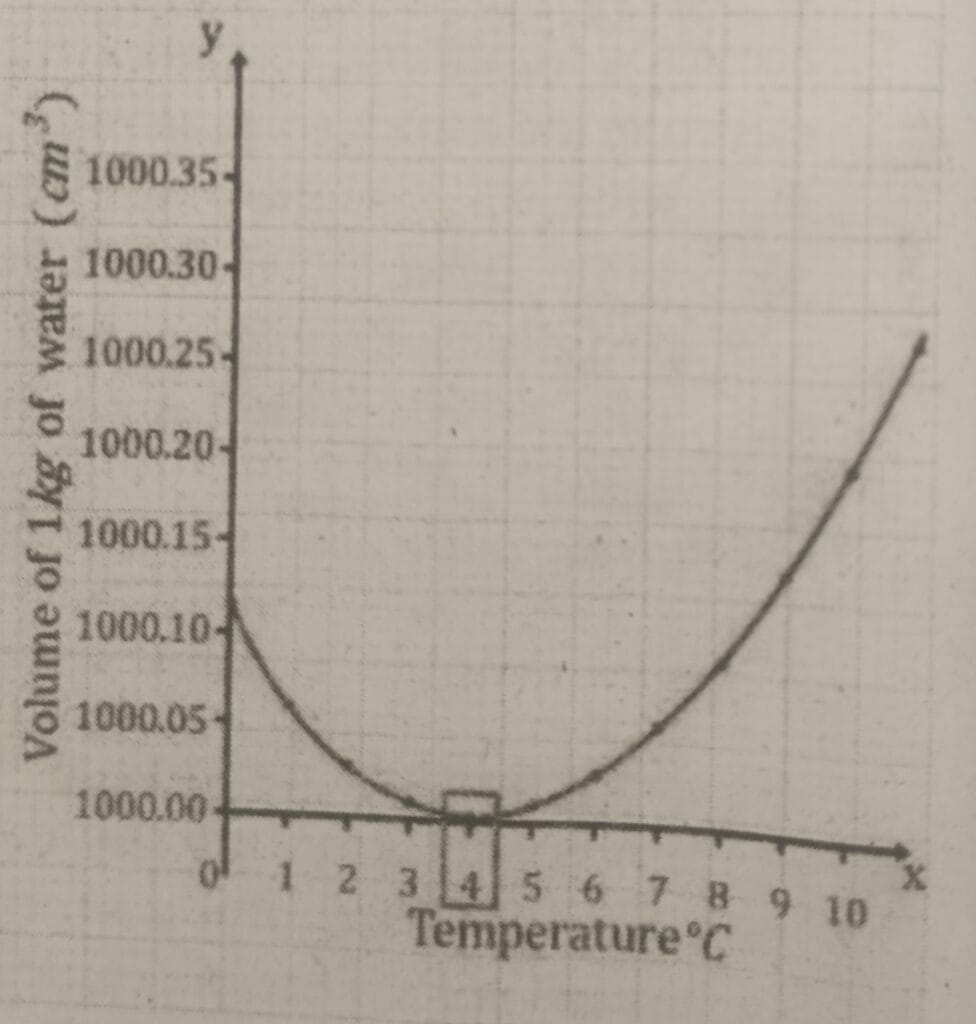
i. What is the special property of water shown in the picture called?
ii. Write the change that took place in density of water on heating water from zero degree to 10⁰C.
iii. Write any one effect of this special property of water.
21. Answer the following questions.
I. Write any two differences between convex lens and concave lens. (U)
ii. Between air and glass, air is called rarer medium and glass is denser medium, why?(U)
22. Answer the following questions on the basis of electronic configuration of element A and B given below. (U)
A = 1s², 2s² 2p⁶, 3s² 3p⁶, 4s¹
B = 1s², 2s² 2p⁴
I. In which group element A belongs in periodic table?
ii. In which period element B belongs in periodic table?
iii. Write down molecular formula of compound formed by combination of A and B.
iV. Why is element B kept in p block of periodic table?
23. Draw available diagram of laboratory preparation of ammonia gas and write down a method to test this gas in the gas there. (A). 1+2+1
Best of Luck
