SEE Practice Model Set 3
[EWA2B3C0D1]
Time: 3 hours FM : 75
Write an answer in your own creative way on the basis of given instructions.
Group ‘A’ [MCQ]
Multiple choice questions. [10X1 =10]
1. Write the best alternative of the following questions in the answer sheet.
a. Which of the following is the function of receiver?
A. To convert received signal into desired form.
B. To receive digital signal from channel.
C. To receive analogue signal from source encoder.
D. To receive signal from source encoder.
b. Why are Moses and ferns both found in moist and sady places?
A. Do not need sunlight for photosynthesis.
B. Survive only at low temperature.
C. Depend on microorganisms for nutrition.
D. Requires water for fertilization.
c. When Sameera observed the specimen in the laboratory, she found that both the organisms were long and cylindrical. She also found that organism A has cylindrical segmented body and organism B has not segmented body. By studying the above characteristics which is statement is true for these organisms?
A. Organism A is diploblastic whereas organism B triploblastic.
B. Organism A is triploblastic whereas organism B is diploblastic.
C. Organism A has well developed circulatory system whereas organism B has no circulatory system.
D. Organism A has no circulatory system whereas B has well developed circulatory system.
d. Which of the following groups is a group of endanger animals?
A. Blackbuck, wild yak, one horned rhinoceros.
B. Brown bear, Bengal tiger, jackal.
C. Red Panda, Pigmy hog, Deer
D. Snow leopard, Asiatic wild elephant, horse.
e. The acceleration due to gravity on the surface of the Earth is g. If mass and radius both are made halved, what is the new value of acceleration due to gravity on the earth surface?
A.g
B. 2g
C. 3g
D. 4g
f. Which principle is the given instrument based on?
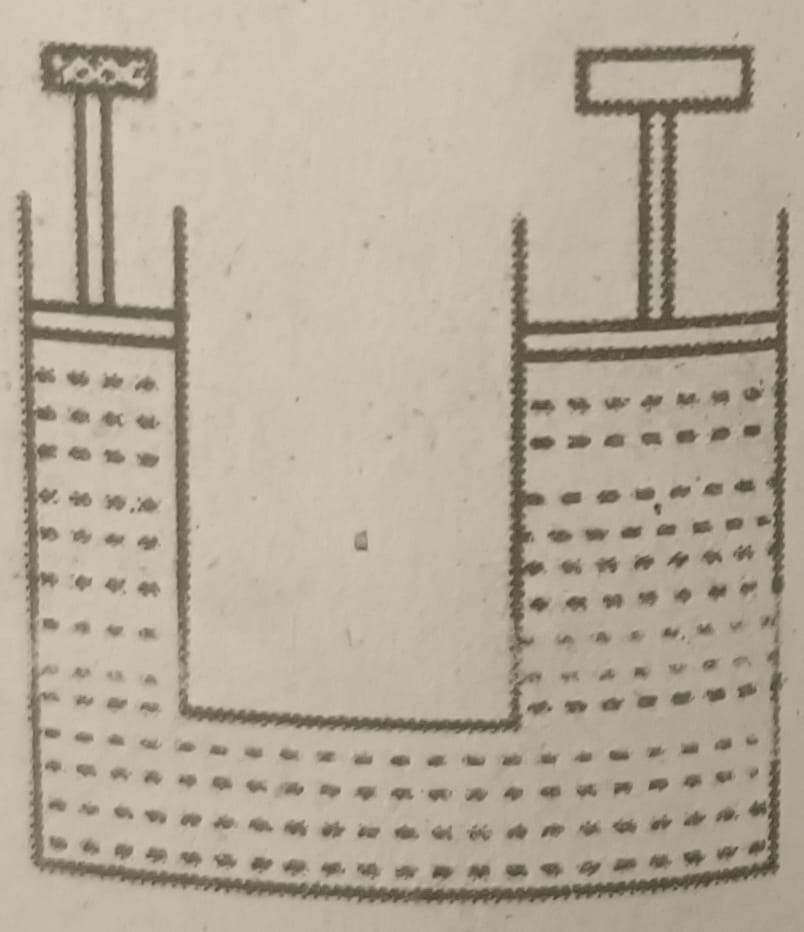
A. Pascal’s law
B. Archimedes’ principle
C. Law of gravitation
D. Newton’s law
g. Soni named the speed of violet, blue, yellow and red coloured beam during the dispersion of light in prism as ev, eb, ey and er respectively. Which is the correct order of a speed made by her?
A. er < ey <eb <ev
B. er > ey > eb > ev
C. er < eb > ey < ev
D. er> eb >ey > ev
h. Under what condition can the flat universe be hypothesized?
A. When average density is equal with a critical density.
B.When average density is less than the critical density.
C. When average density is greater than the critical density.
D. When avarege density and critical density are not compareable.
I. What is X in the given chemical equation?
NaOH + X ➡️ NaCl + H2O
A. NaH
B. Cl2
C. HCl
D. H2O + Cl2
j. Organic matter is considered as a major source of water pollution caused by wastes of food, animal and human excreta, garbage, etc. Which of the following is a threat tqo aquatic life due to the excess of organic matter in water?
A. The lack of space available to aquatic life.
B. Microorganisms consume dissolved oxygen to decompose organic matter.
C. Organic matter is swallowed by small animal.
D. Decomposition of organic matter increases the temperature of water.
Group ‘B’ [9 X 1 = 9]
Very short answers type questions.
a. In an experiment density of water is being measured at different temperature. Which one is dependent variable in this experiment?
b. Write the Archimedes principle.
c. Write a difference between incident ray and emergent ray.
d. Ravi has taken his mother to ophthalmic as he saw reading the newspaper keeping more than 25 cm from her eyes. After the eye examination, what kind of glasses with lenses did the specialist suggest her to wear?
e. What change occur in output voltage if the number of turns in secondary coils is less than the number of turns in primary coil?
f. Write a difference between closed universe and open universe.
g. At what temperature density of water becomes the highest?
h. Why is hematite ore considered chief ore of iron?
I. Write the structural formula of ethanol.
Group ‘C’. [14 X 2 = 28]
Write short answers.
3. Prove that electric resistance ( Ω)= kgm² s–³A–².(A)
4. Write down any two characteristics of the class amphibian. (K)
5. Study the given concept map and name A and B.

6. Write any two differences between somatic chromosome and sex chromosome. (U)
7. Draw a chart of the result obtained by crossing black mouse (BB) and hybrid black mouse(Bb). (A)
8. White blood cell of Renu’s blood is found 2500 after examination her blood. What are the possible causes of this? Write any two causes.(U)
9. Despite the medicinal herbs in our locality, we are dependent apun imported medicines. Write its one cause and what can we do to increase the use of medicinal herbs? Write a point. (A)
10. Write any two effects of gravity. (K)
11. Kavin dropped a stone from a tower of 49 m high. One second later, he throws another stone. They both hit the ground at the same time. Find out the velocity with which he threw the seconds one.(A)
12. Study the given figure and answer the following questions. (U)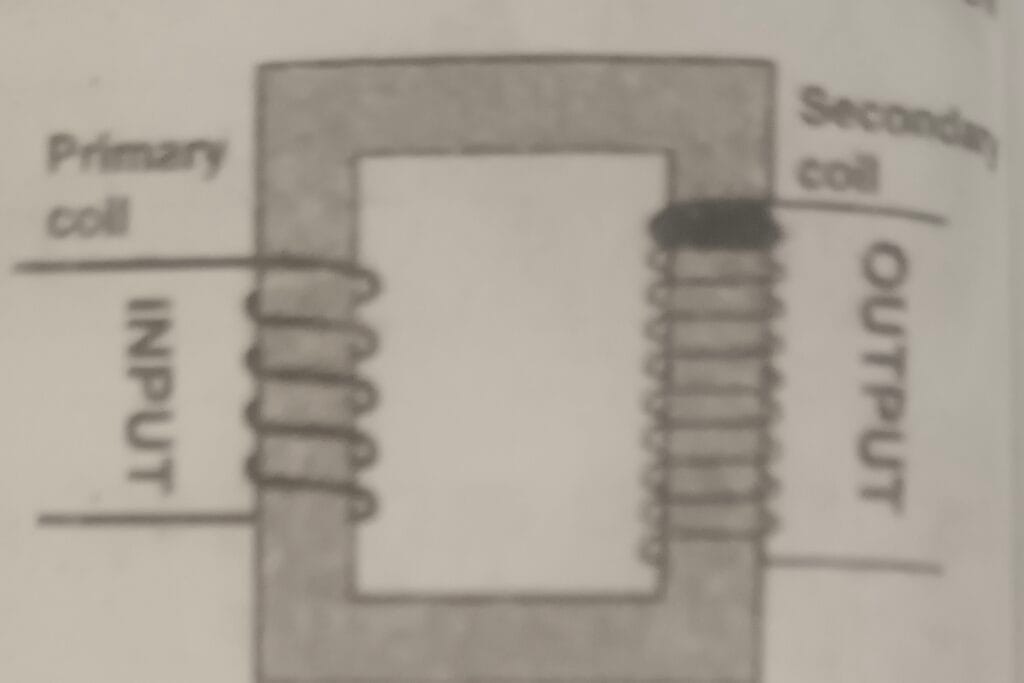 i. What type of transformer is shown in the figure?
i. What type of transformer is shown in the figure?
ii. Why is this used in power station?
13. Niva made the device has shown in figure but rate of rotation of coil is found less. What should be done to increase the rate of rotation of coil? Write any two methods. (A)

14. Suppose equal volume of hydrochloric acid is kept in conical flask A, B and C. If 10 gram powder limestone is kept in A and C, and piece of 10 gram limestone is kept in B, in which conical flask rate of chemical reaction is fast? Write with reason. (HA)

15. The property and use of hydrocarbon compound in the given table below. (HA)
Property | Use |
Colourless and viscous fluid with sweet taste. | Use as sweetening agent in confectionery. |
I. Write down IUPAC name of that compound.
ii. Write down the structural formula of compound formed by reaction between that compound with plenty of hydrogen.
16. A part of opinion of Mr Abhishek harka published in the “My Republic” on 28 April 2023 he is given here ……..” In the Nepal, the use of calcium carbide to ripen fruits prohibited under the food act of 1967. Artificial ripening using chemicals such as calcium carbide can have determinantal effect on human health including poisoning and neurological damage.”
Answer the following questions on the basis of this part. (A)
I. Write down an effect seen in human health except above mentioned by such chemical.
ii. Write a process to ripening food by traditional method.
Group ‘D’
Long answer questions. [7X4=28]
17. Write any two positive and negative impacts of digital technology in our daily life.
18. Study the given figure and answer the following questions.
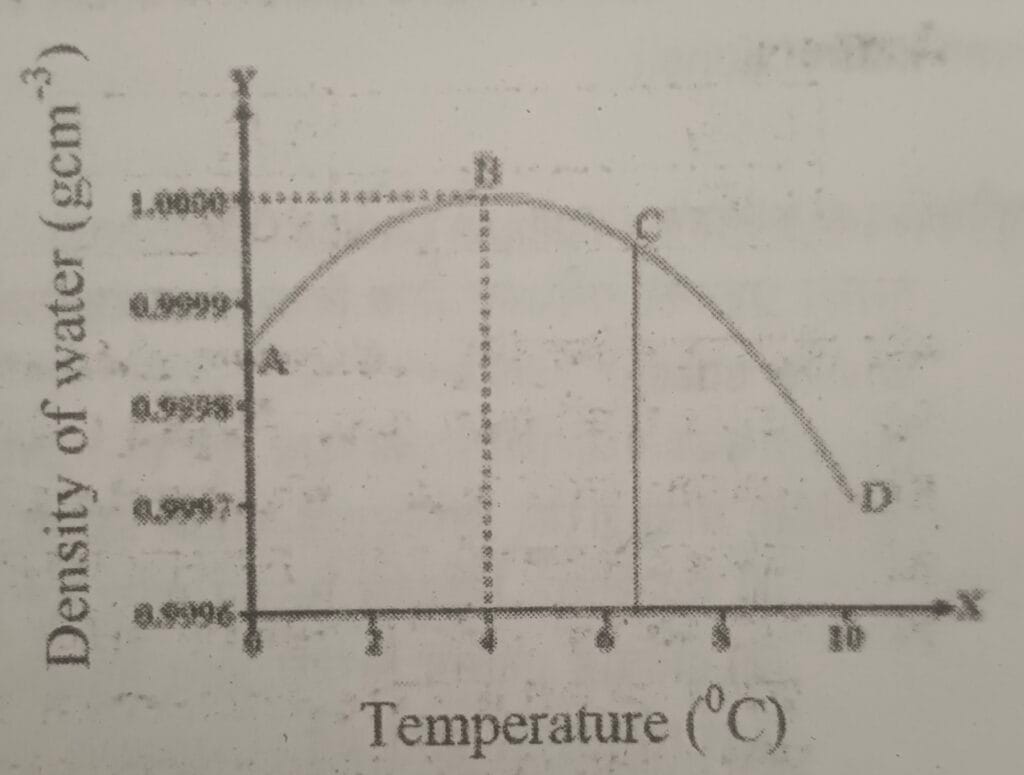
I. What is the special property of water soon in the figure called?
ii. Write the change that takes place in the volume of water on heating from zero degree celsius to 10 degree Celsius.
iii. Write any one use of this special property of water.
19. a. What is an artery? Name the largest artery of human body.
b. Mention two causes of high blood pressure.
20.
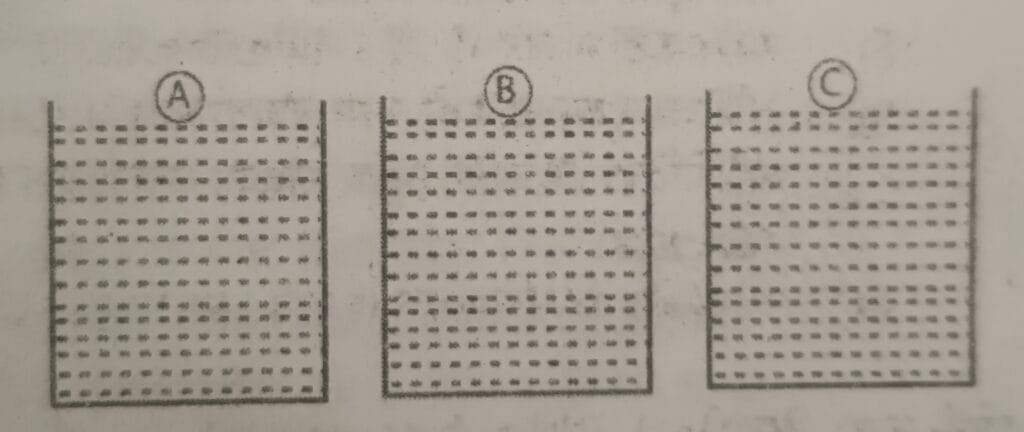
In the figure A, B, and C are three metal objects of equal mass and volume. They are heated to the same temperature and then placed in a vessel containing solid wax. The specific heat capacity of A, B and C are 380 J/kg⁰C, 470J/kg⁰C and 910 J/kg⁰C respectively. Based on this information answer the following questions:
a. Which metal has the highest amount of heat energy?
b. Which metal goes the deepest into the wax vessel?
c. Which metal cools down the fastest?
d. Which metal has the highest amount of heat lose?
21. Write two differences between concave and convex lens.
The power of the lens used in the spectacles worn by a student is -6D. Calculate the focal length of the lens. Also mention the types of lens.
22. Study the electronics configuration of two element X and Y and answer the given questions.
X➡️1s², 2s²2p⁶, 3s²3p⁶, 4s²
Y➡️ 1s², 2s²2p⁶, 3s²3p⁵
a. Which one is metal?
b. Write down the period of element Y.
c. Write the balance chemical equation of the compound formed by the combination of X and Y.
23. Study the given diagram and answer the following questions.
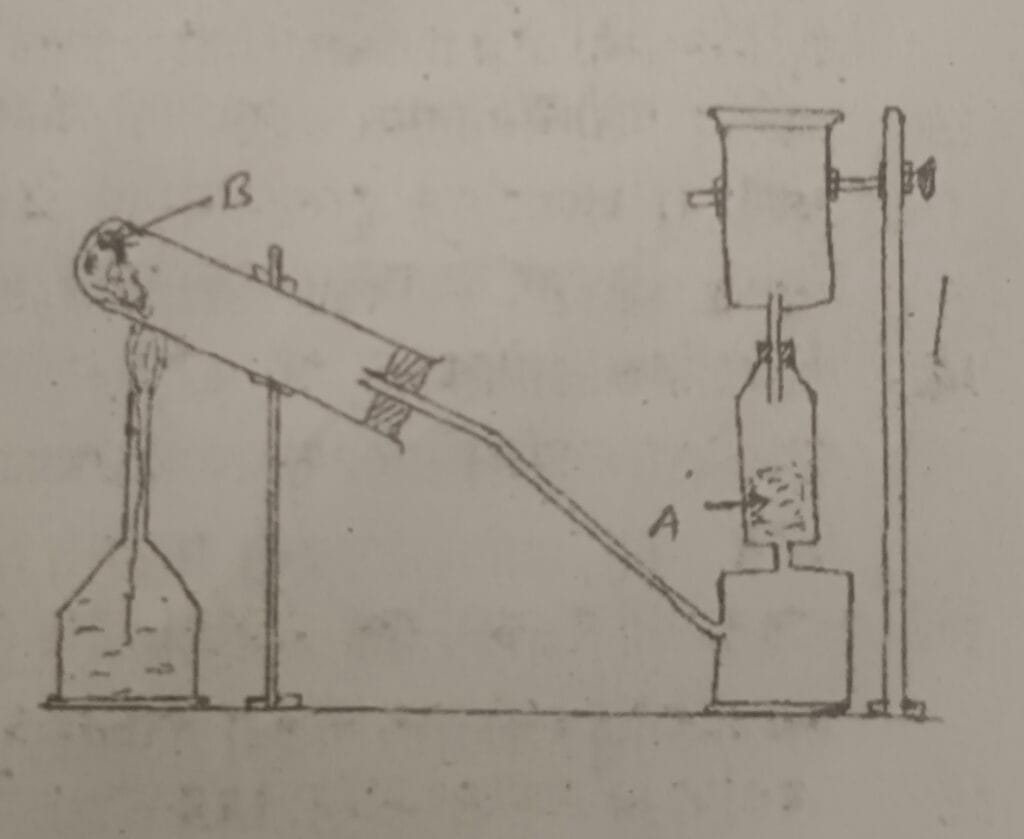
a. Which gas is being collected in the gas jar?
b. What are A & B in the figure?
c. What coloured litmus paper is used to identify such gas?
Best of Luck
