
🔥 1. Heat and Temperature: Summary

🔑 Key Points:
Heat and Temperature
Heat is a form of energy that flows from a hotter body to a colder one.
Unit: Joule (J)
Instrument: Calorimeter
Temperature is the measure of the hotness or coldness of a body.
Unit: Celsius (°C), Fahrenheit (°F), Kelvin (K)
Instrument: Thermometer
Heat and temperature are not the same:
Heat depends on mass and energy.
Temperature depends on the average kinetic energy of particles.
🌡️ 2. Quantity of Heat: Summary

🔑 Key Points:
Quantity of heat (Q) = Heat energy absorbed or given out
Q=mcΔTQ = mcDelta T
mm: mass (kg)
cc: specific heat capacity (J/kg·°C)
ΔTDelta T: change in temperature (°C)
Specific heat capacity is the amount of heat required to raise 1 kg of a substance by 1°C.
Latent heat: heat energy absorbed or released without change in temperature (during melting, boiling)
🔧 3. Thermal Expansion: Summary

🔑 Key Points:
Thermal expansion is the increase in size, area, or volume of a substance due to heat.
Types:
Linear (length)
Areal (surface area)
Volumetric (entire volume)
Solids expand the least, gases the most.
Applications:
Railway gaps
Bimetallic strips
Overhead power lines
🔁 4. Transfer of Heat: Summary
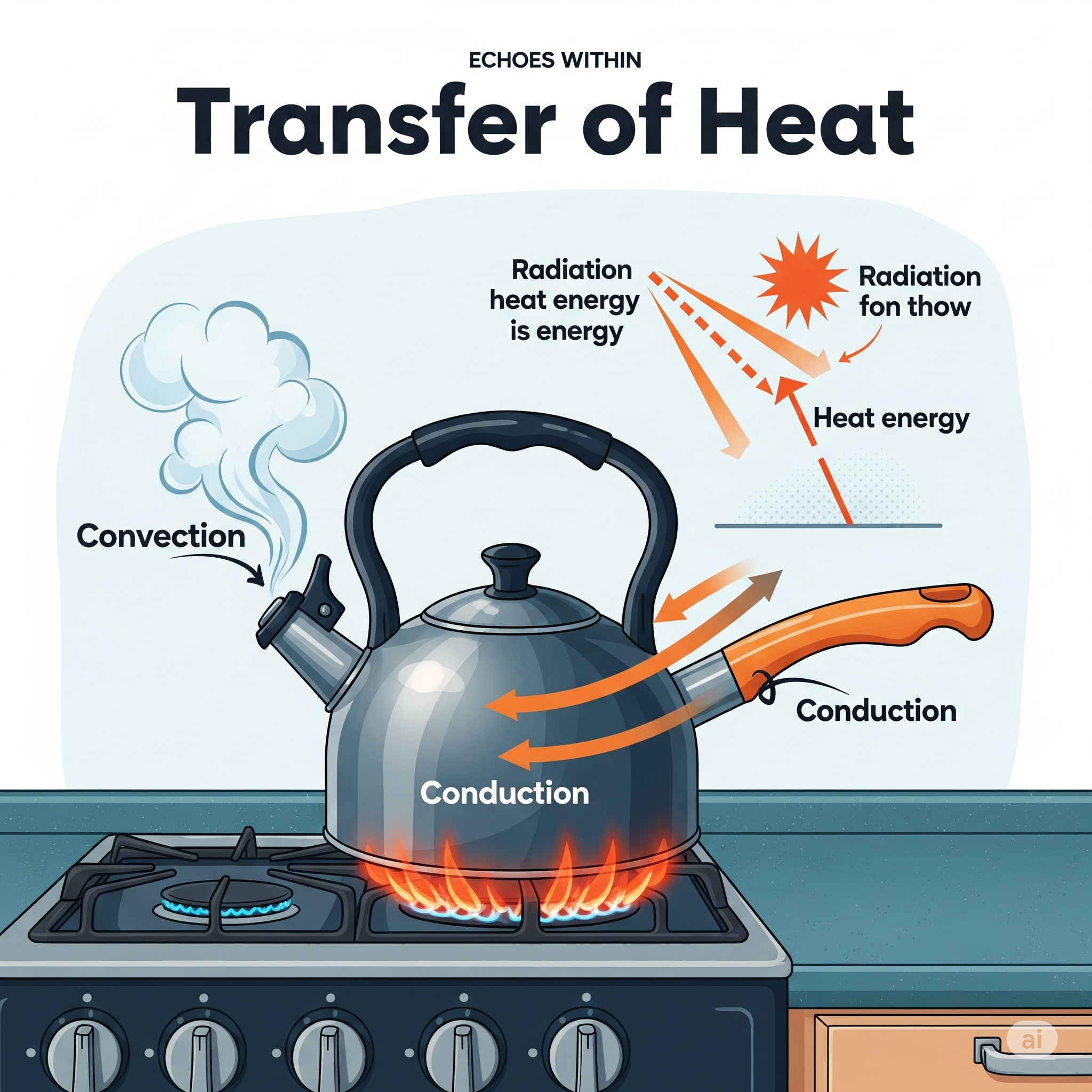
🔑 Key Points:
Heat transfers from hot to cold regions in three ways:
Conduction: through solids (molecule-to-molecule)
Convection: through fluids (liquids & gases by movement)
Radiation: through vacuum (no medium needed)
Good conductors: metals like copper, iron
Poor conductors: wood, plastic, air
Convection examples: sea breeze, boiling water
Radiation example: heat from the Sun
For Further Reading:
📘 Sources of Energy: Chapter Summary for MCQ
You Can Also Read:
The Ethics of Human Genetic Engineering: Playing God or Progress?
Extreme Introversion: Thriving in a World Built for Extroverts
We Are Losing Creativity in the Search for Perfection
